NovoSorb BTM

NovoSorb® BTM (Biodegradable Temporizing Matrix) is a synthetic, biodegradable and biocompatible device designed to facilitate the dermis to grow within a patented polyurethane matrix. When ready, the sealing membrane is removed, leaving a fully vascularized dermis, ready for definitive closure.
NovoSorb BTM is a bilayer matrix

A temporary non-biodegradable layer closes the wound, limiting moisture loss while also serving as a barrier to outside bacteria2,3.
A 2mm bioabsorbable open cell matrix allows for the infiltration of cellular materials and serves as a matrix to aid in the reconstruction of the deeper layers (dermis) of the skin4.
A 2mm bioabsorbable open cell matrix allows for the infiltration of cellular materials and serves as a matrix to aid in the reconstruction of the deeper layers (dermis) of the skin4.
The difference is NovoSorb BTM:
- Robust in the presence of infection – Unlike biologics, NovoSorb BTM’s synthetic composition is not a food supply for bacteria 4
- Designed to minimize scarring and contracture – NovoSorb BTM facilitates the human body to regenerate a neodermis2,5,6
- Cost effective alternative to biologics – typically less than biologic alternatives
- No cold storage – can be stored at room temperature (≤ 25° C)
- Large sizes – up to 20x40cm
- Easy application – cut and apply with sutures or staples
- No tissue tracking – saves data logging time for the clinical team
Indications
NovoSorb® BTM is indicated for use in the management of wounds including: partial and full thickness wounds, pressure ulcers, venous ulcers, diabetic ulcers, chronic and vascular ulcers, surgical wounds (donor sites/grafts, post-Moh’s surgery, post-laser surgery, podiatric, wound dehiscence), trauma wounds (abrasions, lacerations, second-degree burns, and skin tears) and draining wounds.
Phases of NovoSorb BTM Integration



NovoSorb® BTM implanted into a surgically debrided wound bed
The wound is physiologically closed, limiting the risk of infection, evaporative moisture loss and contraction1,2.

Integration process of NovoSorb® BTM
Over a period of approximately 2 to 3 weeks, NovoSorb® BTM integrates into the wound bed through cellular infltration.

NovoSorb® BTM fully integrated
The dermis is regenerated within the matrix. Once fully integrated, the sealing membrane is ready for removal.

Sealing membrane removed
Once the sealing membrane is removed the neodermis is ready for secondary treatment.

Secondary treatment
Method of secondary treatment is left to the physician’s clinical choice (e.g. closure by SSG, or closure by secondary intent). The NovoSorb® BTM progressively biodegrades and is fully absorbed in approximately 18 months7.
Converting wound repair into regeneration
NovoSorb BTM compartmentalizes a large wound into a series of interconnected microwounds. The body easily heals microwounds, promoting organized regenerative healing.
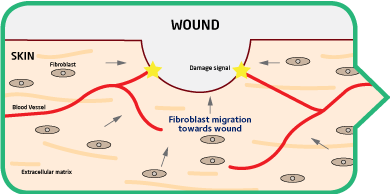
Normal Healing
The body’s natural reparative process follows the chaotic, unorganized laying down of fibrotic tissue in order to rapidly close the wound. This is followed by months of remodeling and scar contraction.


Healing with NovoSorb BTM
NovoSorb BTM provides a unique matrix for organized healing. Cells and blood vessels migrate into the NovoSorb BTM and a new vascularized dermal-like structure is formed5. The body heals each chamber as a discrete small wound8.
NovoSorb BTM Microarchitecture
NovoSorb BTM is an open cell, non-reticulated matrix with interconnected chambers comprised of an estimated 94.2% open space9. This microstructure is stabilized by struts and the chambers are linked by pores, allowing free flow of fluid.
A photomicrograph of NovoSorb
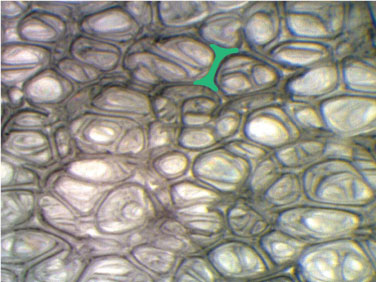
Struts
The solid structural elements of the matrix
Scanning Electron Microscopic

Chamber
The chambers (or cells) highlighted are ~1mm in height
Pores
Smooth, round connections between two chambers. The pores vary in size and average ~188µm10
The NovoSorb BTM chambers compartmentalize the wound, creating microwounds that the body can heal through regeneration. The chambers create a physical barrier that aids in minimizing the foreign body response and helps prevent encapsulation. As healing progresses, a neodermis develops through the matrix.
Cellular Infiltration and Integration through NovoSorb BTM11


When applied, NovoSorb BTM is rapidly infiltrated with hemoserous fluid. As cellular migration begins, the chambers are infiltrated by a variety of cell types with the interconnecting pores7 allowing exchange of nutrients and waste. As healing progresses and a neodermis develops through the matrix, the struts hold their shape, maintaining the microstructure into the remodelling phase.

Day 15
- Cellular activity is seen throughout.
- Light foreign body response is present without encapsulation.
- Fibroblasts commence laying down extracellular matrix with low density after 2 weeks.
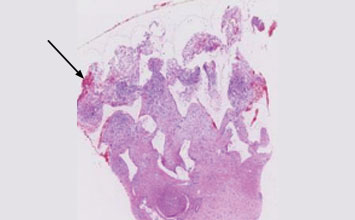
Day 22
- extracellular matrix becomes more dense by 3 weeks.
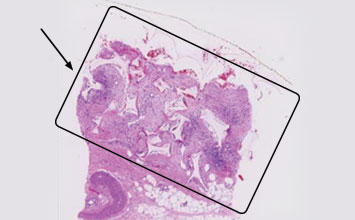
Day 33
- Signifcant dermal integration is achieved.
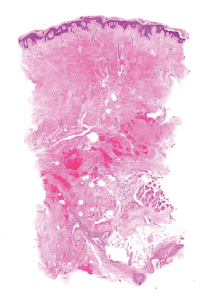
Patient Histology7


NovoSorb BTM is designed to facilitate growth, then safely disappear. NovoSorb BTM gradually Hydrolyzes, shrinking and dissipating until fully reabsorbed in approximately 18 months, leaving a healthy, vascularized neodermis7.
Patient at 7 months
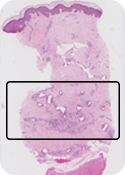
Patient at 8 months


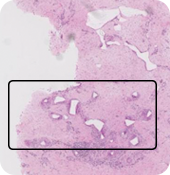
Patient at 12 months
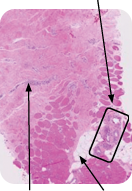
completely gone Fat cells (adipocytes)
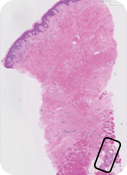
Patient at 18 months
References
- Dearman BL, LiA, Greenwood JE. Optimization of a polyurethane dermal matrix and experience with a polymer-based cultured composite skin. J Burn Care Res. 2014; 35(5): 437-48.
- Greenwood JE, Dearman BL. Comparison of a sealed, polymer foam biodegradable temporising matrix against Integra(R) dermal regeneration template in a porcine wound model. J Burn Care Res. 2012; 33:163-73.
- Wagstaff MJD, Schmitt B, Caplash Y, Greenwood JE. Free flap donor site reconstruction: A prospective case series using an optimized polyurethane temporising matrix. Eplasty. 2015; 15:231-48.
- Greenwood JE, Schmitt BJ, Wagstaff MJD. Experience with a synthetic bilayer Biodegradable temporising Matrix in significant burn injury. Burns Open. 2018;2(1):17-34.
- Greenwood JE, Dearman BL. Split skin graft application over an integrating, biodegradable temporising polymer matrix: immediate and delayed. J Burn Care Res. 2012; 33:7-19.
- Wagstaff MJD, Slana IM, Caplash Y, Greenwood JE. Biodegradable temporising Matrix (BTM) for the reconstruction of defects following serial debridement for necrotising fasciitis: A case series. Burns Open. 2019; 3:12-30.
- Wagstaff MJD, Schmitt BJ, Coghlan P, Finkemeyer JP, Caplash Y, Greenwood JE. A biodegradable polyurethane dermal matrix in reconstruction of free flap donor sites: a pilot study. ePlasty 2015; 15:102-18.
- Greenwood JE, Wagstaff MJD (2016) The use of biodegradable polyurethane in the development of dermal scaffolds. In: Cooper SL, Guan J (eds) Advances in Polyurethane Materials. 1st edn. Woodhead Publishing, pp 631-62.
- Data on file TD-114.
- Internal testing.
- Data on file: Punch biopsies.


