Clinical Application
Clinical Application
NovoSorb® BTM is convenient to store and prepare for application:
- Provided in a multilayer packaging, it can be stored at room temperature (≤ 25° C)
- Cut to wound size, no need for reconstituting or other pre-treatment
- It does not contain any biological materials or sensitizing proteins, so does not require tissue tracking
* Refer to disclaimer below.
Two key stages with NovoSorb BTM:
Application
Once the wound is prepared, NovoSorb BTM is stapled or sutured in place with the device flush against the edges of the wound and the NovoSorb BTM laid flat across the wound bed without creases. The NovoSorb BTM is left in place with regular overlying dressing changes.
Delamination
Once capillary refll is seen throughout the NovoSorb BTM, the sealing membrane is delaminated with the top layer detaching with a ‘Velcro®-like’ action.
Guidelines for the Application and Management of NovoSorb BTM
The following overview is meant to serve as a general guideline. Please refer to the Instructions For Use (IFU) for full instructions. These guidelines are not designed to replace existing institutional protocols or professional clinical judgment regarding patient care.
-
Section 1: Wound Preparation
NovoSorb BTM is designed to integrate to the wound bed when the margins and bed consist of viable tissue.
Preparing the Wound Bed
- Completely excise all non-viable tissue in the wound
- Hemostasis should be achieved in the wound bed
- The wound should be free of infection
-
Section 2: Preparing NovoSorb BTM
Preparing the NovoSorb BTM
- Open the package on the “chevron” side (the tip of the arrow-shaped seal)
- NovoSorb BTM is supplied pre-fenestrated to allow the drainage of exudate. If the wound bed is expected to be heavily exudative, the sealing membrane may be further fenestrated with a scalpel to provide additional drainage holes
- NovoSorb BTM is ready for application straight from the packaging, no further preparation is required.
- Using surgical scissors, cut NovoSorb BTM to the appropriate size
Note: Do not mesh NovoSorb BTM.
-
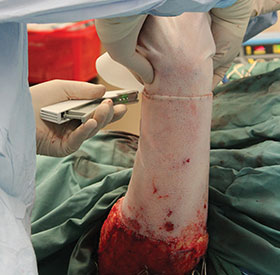
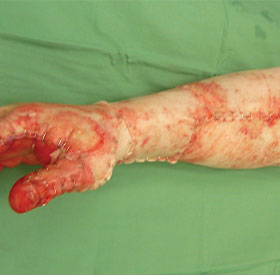
Section 3: Applying NovoSorb BTM
Applying NovoSorb BTM to the Wound Bed
- Apply NovoSorb BTM with porous side in contact with wound bed. Note, the porous side may sparkle under surgical lights.
- NovoSorb BTM should lay flat in the wound bed with no creases
- Once flat against the wound bed, NovoSorb BTM is secured with staples or sutures
Note
- For large areas of application, quilting staples can be used to secure the NovoSorb BTM to the wound bed
- Adjacent pieces of NovoSorb BTM can be stapled or sutured together as required
- Avoid overlapping adjacent pieces
-
Section 4: Managing Outer Dressings
Outer Dressings
The choice of outer dressings should:
- Not adhere to NovoSorb BTM
- Absorb mild exudate
- Possess an antimicrobial property
- Encourage continued contact of the NovoSorb BTM to the wound bed without shear
Collections of turbid fluid or hematomas under the seal can be expressed via the fenestrations in the overlying seal. If necessary, the sealing membrane may be further fenestrated with a scalpel to provide drainage holes.
For example:
- Apply a non-adherent contact layer directly over NovoSorb BTM
- Apply an antimicrobial dressing
- Apply gauze or other absorbent dressing
- Apply outer crepe dressing

-
Section 5: Clinical Application
Performing Wound Care
Under aseptic conditions:
- Remove all dressings; non-adherent contact layer may be retained or discarded at frst dressing change per clinical judgment
- Do not remove NovoSorb BTM and/or staples/sutures
- Gently wipe the NovoSorb BTM surface with a saline gauze and/or antimicrobial preparation such as chlorhexidine solution or povidone-iodine solution
- Replace dressings following standard of care
The dressings should be removed and replaced as frequently as required for:
- Dressings with absorbed exudate to be discarded
- Replacement of antibacterial agents once their period of therapeutic effect has expired
- Staples or sutures remain in place until NovoSorb BTM is fully integrated to the wound or until delamination.

-
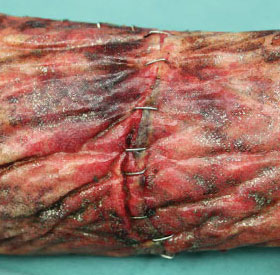
Section 6: Assessing Tissue Integration
Week 1–2 Tissue Integration
- NovoSorb BTM matrix is visible through the clear sealing membrane
- NovoSorb BTM is dark red in color due to ingress of blood from the wound
- NovoSorb BTM gradually changes to a lighter red color during this time period
- Collections of turbid fluid or hematomas under the seal can be expressed via the fenestrations in the overlying seal
- By the end of week 2, signs of integration should be visible
- Once NovoSorb BTM has adhered to the wound bed, range of motion can begin
Note: The color of the NovoSorb BTM at various stages of integration may vary between patients.
Week 3-6 Tissue Integration
- The microstructure and patterns seen initially through the seal are gradually obliterated as the NovoSorb BTM further integrates
- Later during this time period capillary refill (blanching) may be observed by applying digital pressure to the sealing membrane
- When the microstructure is no longer visible, there is capillary refill and the matrix is a uniform pink color, then the NovoSorb BTM is ready for delamination
Note: The use of certain silver dressings may leave a black tarnished appearance on the surface of the NovoSorb BTM.
-
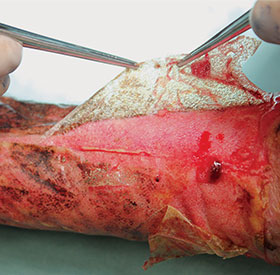
Section 7: Delaminating BTM
Delaminating the NovoSorb BTM
- Remove all staples and/or sutures
- Using forceps, raise a free edge of the sealing membrane
- Peel the membrane off the matrix using gentle, even traction
Note: Any unintegrated matrix fragments will pull away with the seal. However, integrated NovoSorb BTM will remain firmly adhered to the wound bed.
-
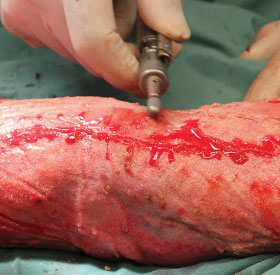
Section 8: Wound Closure Options
Methods of Wound Closure
It is clinician’s choice to determine best method of wound closure for their patient. Options include:
Split Thickness Skin Graft
- Prepare skin graft for closure following standard protocol
- Skin grafts can be applied and dressed according to standard of care
Note: A mild refresher to the newly delaminated surface may be considered.1,2
Secondary Intention
- For wound closure by secondary intention healing, the integrated and delaminated NovoSorb BTM can be dressed according to standard of care.

References
- Greenwood et al, Reconstruction of Extensive Calvarial Exposure After Major Burn Injury in 2 Stages Using a Biodegradable Polyurethane Matrix, Eplasty, Vol.16, ID e17, 2016.
- Wagstaff et al, Reconstruction of an Anterior Cervical Necrotizing Fasciitis Defect Using a Biodegradable Polyurethane Dermal Substitute, Eplasty, Vol. 17, ID e3, 2017.
Velcro® is a registered trademark of Velcro BVBA.
Disclaimer
The information contained here are exclusively aimed at health care professionals.
Always read the label and follow the instructions for use including the contraindications, warnings, and precautions.
As with any surgical procedure, this treatment has potential benefits and risks. It is important to understand these potential benefits and risks when considering the treatment options for closing the wound and aid the body in generating new tissue.





